Tissue factor Anticorps
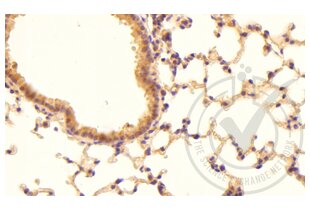
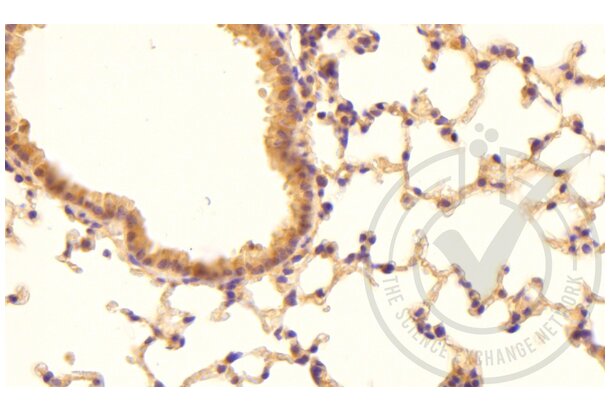 Tissue factor anticorps (AA 32-100) (ABIN708086)
Tissue factor anticorps (AA 32-100) (ABIN708086)
F3 Reactivité: Humain, Souris, Rat, Porc WB, ELISA, FACS, IHC (p), IF (cc), IF (p), IHC (fro) Hôte: Lapin Polyclonal unconjugated
F3 Reactivité: Humain WB, ELISA, IP, PLA Hôte: Souris Monoclonal 4G4 unconjugated
F3 Reactivité: Humain WB, ICC, FACS, IHC (p) Hôte: Souris Monoclonal HTF-1 unconjugated
Tissue factor Anticorps par Réactivité
Trouvez Tissue factor Anticorps pour une variété d'espèces telles que anti-Human Tissue factor, anti-Mouse Tissue factor, anti-Rat Tissue factor. Les espèces listées ci-dessous sont parmi celles disponibles. Cliquez sur un lien pour accéder aux produits correspondants.
Tissue factor Anticorps par Hote
On trouve ici des Tissue factor Anticorps avec un Hote spécifique. Les Hote mentionnés ici sont quelques-uns de ceux qui sont disponibles. Un clic sur le lien correspondant permet d'accéder aux produits
Tissue factor Anticorps par Clonalité
Trouvez les Tissue factor Anticorps monoclonaux ou polyclonaux disponibles. Cliquez sur un lien pour accéder aux produits correspondants.
Tissue factor Anticorps par Clone
On trouve ici des Tissue factor Anticorps avec un Clone spécifique. Les Clone mentionnés ici sont quelques-uns de ceux qui sont disponibles. Un clic sur le lien correspondant permet d'accéder aux produits
Tissue factor Anticorps par Fragment
On trouve ici des Tissue factor Anticorps avec un Fragment spécifique. Les Fragment mentionnés ici sont quelques-uns de ceux qui sont disponibles. Un clic sur le lien correspondant permet d'accéder aux produits
Tissue factor Anticorps fréquemment utilisés
- (7)
- (6)
- (1)
- (7)
- (5)
- (5)
- (5)
- (1)
- (4)
- (4)
- (3)
- (3)
- (3)
- (2)
- (2)
- (2)
- (2)
- (2)
Dernières publications pour nos Tissue factor Anticorps
: "Participation of Extracellular Vesicles from Zika-Virus-Infected Mosquito Cells in the Modification of Naïve Cells' Behavior by Mediating Cell-to-Cell Transmission of Viral Elements." dans: Cells, Vol. 9, Issue 1, (2020) (PubMed).: "CTNNB1/β-catenin dysfunction contributes to adiposity by regulating the cross-talk of mature adipocytes and preadipocytes." dans: Science advances, Vol. 6, Issue 2, pp. eaax9605, (2020) (PubMed).
: "Protease-activated receptor 2 protects against VEGF inhibitor-induced glomerular endothelial and podocyte injury." dans: Scientific reports, Vol. 9, Issue 1, pp. 2986, (2019) (PubMed).
: "Identification of a mesenchymal progenitor cell hierarchy in adipose tissue." dans: Science (New York, N.Y.), Vol. 364, Issue 6438, (2019) (PubMed).
: "Effect of MSCs and MSC-Derived Extracellular Vesicles on Human Blood Coagulation." dans: Cells, Vol. 8, Issue 3, (2019) (PubMed).
: "Super-Enhancers Promote Transcriptional Dysregulation in Nasopharyngeal Carcinoma." dans: Cancer research, Vol. 77, Issue 23, pp. 6614-6626, (2018) (PubMed).
: "Chimeric antigen receptor-modified T Cells inhibit the growth and metastases of established tissue factor-positive tumors in NOG mice." dans: Oncotarget, Vol. 8, Issue 6, pp. 9488-9499, (2018) (PubMed).
: "The Low Molecular Weight Heparin Tinzaparin Attenuates Platelet Activation in Terms of Metastatic Niche Formation by Coagulation-Dependent and Independent Pathways." dans: Molecules (Basel, Switzerland), Vol. 23, Issue 11, (2018) (PubMed).
: "Endothelial cell functions impaired by interferon in vitro: Insights into the molecular mechanism of thrombotic microangiopathy associated with interferon therapy." dans: Thrombosis research, Vol. 163, pp. 105-116, (2018) (PubMed).
: "Androgen receptor dampens tissue factor expression via nuclear factor-κB and early growth response protein 1." dans: Journal of thrombosis and haemostasis : JTH, Vol. 16, Issue 4, pp. 749-758, (2018) (PubMed).
Pseudonymes pour Tissue factor Anticorps
coagulation factor III, tissue factor (F3) Anticorpstransferrin (TF) Anticorps
coagulation factor IIIa (f3a) Anticorps
coagulation factor III (F3) Anticorps
tissue factor (tf) Anticorps
coagulation factor IIIb (f3b) Anticorps
coagulation factor III (thromboplastin, tissue factor) S homeolog (f3.S) Anticorps
AA409063 Anticorps
CD142 Anticorps
Cf-3 Anticorps
Cf3 Anticorps
f3 Anticorps
PRO1557 Anticorps
PRO2086 Anticorps
TF Anticorps
tf Anticorps
TFA Anticorps
TFQTL1 Anticorps
zgc:112151 Anticorps
Avez-vous cherché autre chose?
- TIRAP Anticorps
- TIPRL Anticorps
- TIPIN Anticorps
- Tiparp Anticorps
- TINF2 Anticorps
- TINAGL1 Anticorps
- TINAG Anticorps
- TIMP4 Anticorps
- TIMP3 Anticorps
- TIMP1 Anticorps
- TIMM9 Anticorps
- TIMM8B Anticorps
- TIMM8A/DDP Anticorps
- TIMM50 Anticorps
- TIMM44 Anticorps
- TIMM29 Anticorps
- TIMM23 Anticorps
- TIMM22 Anticorps
- TIMM21 Anticorps
- TIMM17B Anticorps




