DLG4 Anticorps
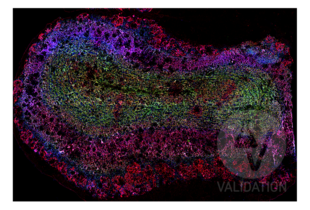
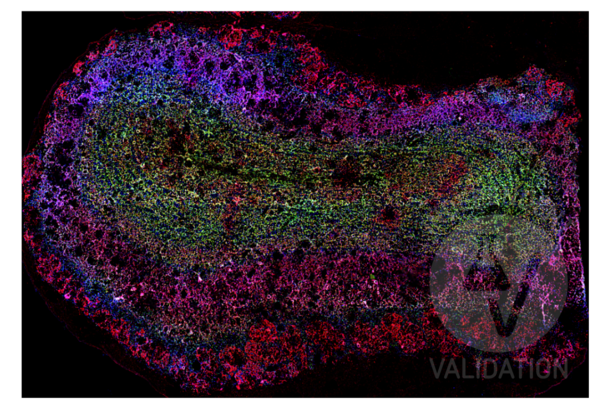 DLG4 anticorps (ABIN361694)
DLG4 anticorps (ABIN361694)
DLG4 Reactivité: Rat WB, IHC, IF, ICC, AA, mIHC Hôte: Souris Monoclonal 6G6 unconjugated
DLG4 Reactivité: Rat WB, IHC, IF, ICC, AA Hôte: Souris Monoclonal 7E3 unconjugated
DLG4 Reactivité: Rat WB, IHC, IF, ICC, AA Hôte: Souris Monoclonal 7E3 HRP
DLG4 Anticorps par Grade
On trouve ici des DLG4 Anticorps avec un Grade spécifique. Les Grade mentionnés ici sont quelques-uns de ceux qui sont disponibles. Un clic sur le lien correspondant permet d'accéder aux produits
DLG4 Anticorps par Hote
On trouve ici des DLG4 Anticorps avec un Hote spécifique. Les Hote mentionnés ici sont quelques-uns de ceux qui sont disponibles. Un clic sur le lien correspondant permet d'accéder aux produits
DLG4 Anticorps par Clonalité
Trouvez les DLG4 Anticorps monoclonaux ou polyclonaux disponibles. Cliquez sur un lien pour accéder aux produits correspondants.
DLG4 Anticorps fréquemment utilisés
- (3)
- (1)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (4)
- (5)
- (4)
- (3)
Dernières publications pour nos DLG4 Anticorps
: "Ginsenoside Rg1 alleviates Aβ deposition by inhibiting NADPH oxidase 2 activation in APP/PS1 mice." dans: Journal of ginseng research, Vol. 45, Issue 6, pp. 665-675, (2021) (PubMed).: "Histological Correlates of Neuroanatomical Changes in a Rat Model of Levodopa-Induced Dyskinesia Based on Voxel-Based Morphometry." dans: Frontiers in aging neuroscience, Vol. 13, pp. 759934, (2021) (PubMed).
: "Electroacupuncture Ameliorates Depression-Like Behaviour in Rats by Enhancing Synaptic Plasticity via the GluN2B/CaMKII/CREB Signalling Pathway." dans: Evidence-based complementary and alternative medicine : eCAM, Vol. 2021, pp. 2146001, (2021) (PubMed).
: "Essential omega-3 fatty acids tune microglial phagocytosis of synaptic elements in the mouse developing brain. ..." dans: Nature communications, Vol. 11, Issue 1, pp. 6133, (2020) (PubMed).
: "Adolescent social instability stress alters markers of synaptic plasticity and dendritic structure in the medial amygdala and lateral septum in male rats." dans: Brain structure & function, Vol. 224, Issue 2, pp. 643-659, (2019) (PubMed).
: "The effects of social instability stress and subsequent ethanol consumption in adolescence on brain and behavioural development in male rats." dans: Alcohol (Fayetteville, N.Y.), (2019) (PubMed).
: "NF-κB-regulated microRNA-574-5p underlies synaptic and cognitive impairment in response to atmospheric PM2.5 aspiration." dans: Particle and fibre toxicology, Vol. 14, Issue 1, pp. 34, (2018) (PubMed).
: "Dcf1 Triggers Dendritic Spine Formation and Facilitates Memory Acquisition." dans: Molecular neurobiology, Vol. 55, Issue 1, pp. 763-775, (2018) (PubMed).
: "Sex-specific effects of CB1 receptor antagonism and stress in adolescence on anxiety, corticosterone concentrations, and contextual fear in adulthood in rats." dans: International journal of developmental neuroscience : the official journal of the International Society for Developmental Neuroscience, Vol. 69, pp. 119-131, (2018) (PubMed).
: "Age-related effects of X-ray irradiation on mouse hippocampus." dans: Oncotarget, Vol. 7, Issue 19, pp. 28040-58, (2018) (PubMed).
Pseudonymes pour DLG4 Anticorps
discs large 1 (dlg1) Anticorpsdiscs, large homolog 4 (dlg4) Anticorps
discs large MAGUK scaffold protein 4 (DLG4) Anticorps
discs, large homolog 4b (Drosophila) (dlg4b) Anticorps
discs, large homolog 4 (Drosophila) (Dlg4) Anticorps
discs large MAGUK scaffold protein 4 (Dlg4) Anticorps
11 Anticorps
anon-EST:Posey93 Anticorps
anon-WO03040301.258 Anticorps
anon-WO03040301.260 Anticorps
anon-WO03040301.268 Anticorps
CG1725 Anticorps
CG1730 Anticorps
CPD Anticorps
d. lg.-1 Anticorps
Discs-large Anticorps
DLG Anticorps
Dlg Anticorps
dlg Anticorps
dlg-1 Anticorps
DLG-A Anticorps
Dlg-A Anticorps
dlg-A Anticorps
Dlg1 Anticorps
DLG4 Anticorps
DlgA Anticorps
dlgA Anticorps
dlgh4 Anticorps
Dlgh4 Anticorps
dlgS97 Anticorps
Dmel\\CG1725 Anticorps
Drodlg Anticorps
l(1)10Bf Anticorps
l(1)bwn Anticorps
l(1)d.lg-1 Anticorps
l(1)d.lg.-1 Anticorps
l(1)discs large Anticorps
l(1)dlg Anticorps
l(1)dlg-1 Anticorps
l(1)dlg1 Anticorps
l(1)G0276 Anticorps
l(1)G0342 Anticorps
l(1)G0456 Anticorps
l(1)G19 Anticorps
l(1)l.pr.-2 Anticorps
l(1)L11 Anticorps
l(1)lpr-2 Anticorps
LLGL1 Anticorps
misb Anticorps
PSD-95 Anticorps
PSD95 Anticorps
psd95 Anticorps
sap-90 Anticorps
SAP-90 Anticorps
sap90 Anticorps
SAP90 Anticorps
Sap90 Anticorps
SAP90A Anticorps
SAP97 Anticorps
Avez-vous cherché autre chose?
- DLG3 Anticorps
- DLG2 Anticorps
- DLG1 Anticorps
- DLEC1 Anticorps
- DLD Anticorps
- DLC1 Anticorps
- DLAT Anticorps
- DKK4 Anticorps
- DKK3 Anticorps
- DKK2 Anticorps
- DKK1 Anticorps
- DKC1 Anticorps
- DIXDC1 Anticorps
- Distal-Less Homeobox 5 Anticorps
- Distal-Less Homeobox 4 Anticorps
- DISP2 Anticorps
- DISP1 Anticorps
- Disembodied Anticorps
- DISC1 Anticorps
- DIS3L2 Anticorps
- DLG5 Anticorps
- DLGAP1 Anticorps
- DLGAP2 Anticorps
- DLGAP3 Anticorps
- DLGAP4 Anticorps
- DLGAP5 Anticorps
- DLK1 Anticorps
- DLK2 Anticorps
- DLL1 Anticorps
- DLL3 Anticorps
- DLL4 Anticorps
- DLST Anticorps
- DLX1 Anticorps
- DLX2 Anticorps
- DLX3 Anticorps
- DLX6 Anticorps
- DMAP1 Anticorps
- DMBT1 Anticorps
- DMBX1 Anticorps
- DMC1 Anticorps




