P27 anticorps
-
- Antigène
- P27
-
Reactivité
- Humain, Souris, Rat, Singe
-
Hôte
- Souris
-
Clonalité
- Monoclonal
-
Conjugué
- Inconjugué
-
Application
- Western Blotting (WB), Immunohistochemistry (Paraffin-embedded Sections) (IHC (p)), Flow Cytometry (FACS), Immunofluorescence (IF)
- Purification
- Protein G affinity chromatography
- Immunogène
- Mouse recombinant protein (DCS-72.F6) and recombinant human protein (KIP1/769) were used as the immunogen for the p27 antibody cocktail.
- Clone
- DCS-72-F6-KIP1-769
- Isotype
- IgG
-
-
- Indications d'application
-
Optimal dilution of the p27 antibody to be determined by the researcher.
1. Staining of formalin-fixed tissues requires boiling tissue sections in 10 mM Citrate buffer, pH 6.0, for 10-20 min followed by cooling at RT for 20 min
2. The prediluted format is supplied in a dropper bottle and is optimized for use in IHC. After epitope retrieval step (if required), drip mAb solution onto the tissue section and incubate at RT for 30 min.\. Flow Cytometry: 0.5-1 μg/million cells in 0.1ml,Immunofluorescence: 0.5-1 μg/mL,Western blot: 0.5-1 μg/mL,Immunohistochemistry (FFPE): 0.25-0.5 μg/mL for 30 min at RT (1),Prediluted format : incubate for 30 min at RT (2) - Restrictions
- For Research Use only
-
- by
- Johann-Friedrich-Blumenbach-Institute for Zoology and Anthropology, Department of Developmental Biology, Georg-August-University Göttingen
- No.
- #101901
- Date
- 04.12.2017
- Antigène
- P27
- Numéro du lot
- V2438-171009
- Application validée
- Western Blotting
- Contrôle positif
- NIH/3T3 mouse embryonic fibroblast cells overexpressing mVenus-tagged p27K, starved for 48h
- Contrôle négative
- Untransfected NIH/3T3 cells, starved for 48h
- Conclusion
Passed. ABIN3025539 detects the ectopically expressed fusion protein as well as the endogenous protein by immunoblotting. Unspecific cross-reactivity is low.
- Anticorps primaire
- ABIN3025539
- Anticorps secondaire
- anti-mouse IgG (whole molecule), HRP-linked (Sigma-Aldrich, A9044, lot 034M4761)
- Full Protocol
- Grow NIH/3T3 cells (ATCC, CRL-1658) in DMEM+GlutaMAX (Gibco, 31966-021, Lot 1852045) supplemented with fetal bovine serum (Gibco 270-106) and Pen/Strep (Gibco 15140), at 37°C and 5% CO2 to 70% confluency.
- Transfect cells with a plasmid encoding mVenus-tagged p27K- (kindly provided by Toshio Kitamura, University of Tokyo; Oki et al., 2014) using EndofectinMax (GeneCopoeia) following the manufacturer's instructions.
- Serum starve cells for 48h. Use untransfected NIH/3T3 cells starved for 48h as control.
- Lyse cells in RIPA buffer (10mM PBS pH7.2, 2mM EDTA, 1% NP-40, 1% Triton X-100, protease inhibitors) at 4°C.
- Denature total cellular lysates proteins in 1x SDS-sample buffer and separate proteins on a freshly cast denaturing 10% SDS-PAGE (Laemmli, 1970).
- Transfer proteins onto 0.2µm Protran membrane (GE Healthcare, 10600004, A10043108) with a Western blotting system for 1h at 400A (Towbin et al., 1979).
- Block the membrane in TBST (50mM Tris-HCl, pH7.4, 150mM NaCl, 0.2% Tween 20) containing 5% milk (blocking solution) for 60min at RT.
- Incubate membrane with primary mouse anti-P27 antibody (antibodies-online, ABIN3025539, lot V2438-171009) diluted 1:250 in blocking solution ON at 4°C.
- Incubate membrane with secondary anti-mouse IgG (whole molecule), HRP-linked (Sigma-Aldrich, A9044, lot 034M4761) diluted 1:5000 in TBST for 45min at RT.
- Wash membrane with TBST for 30-45min at RT.
- Reveal protein bands using Clarity Max Western ECL substrate (Bio-Rad, 1705062); image capture via Chemidoc Imaging System (BioRad).
- Notes
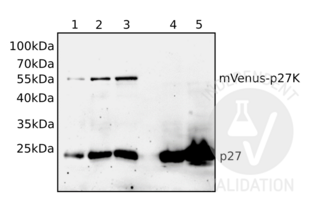
ABIN3025539 reveals a protein band at the expected molecular weight of the mVenus-p27 fusion protein. The antibody does also detect a protein at the expected molecular weight of the endogenous, untagged p27.
Validation #101901 (Western Blotting)![Testé avec succès 'Independent Validation' signe]()
![Testé avec succès 'Independent Validation' signe]() Validation Images
Validation Images![Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.]() Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.
Protocole
Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.
Protocole -
- by
- Johann-Friedrich-Blumenbach-Institute for Zoology and Anthropology, Department of Developmental Biology, Georg-August-University Göttingen
- No.
- #102346
- Date
- 04.12.2017
- Antigène
- P27
- Numéro du lot
- V2438-171009
- Application validée
- Immunocytochemistry
- Contrôle positif
- NIH/3T3 mouse embryonic fibroblast cells overexpressing mVenus-tagged p27K, starved for 48h
- Contrôle négative
- Untransfected NIH/3T3 cells, starved for 48h
- Conclusion
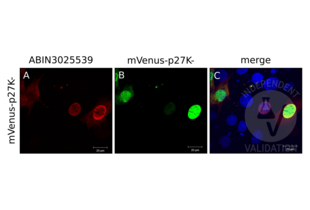
Passed. ABIN3025539 detects the ectopically expressed fusion protein by immunocytochemistry. Unspecific cross-reactivity is low.
- Anticorps primaire
- ABIN3025539
- Anticorps secondaire
- goat anti-mouse IgG (H+L) Alexa Fluor 555 (Invitrogen, A21422, lot 948498)
- Full Protocol
- Grow NIH/3T3 cells (ATCC, CRL-1658) in on cover slips in DMEM, 10% fetal bovine serum (Gibco 270-106), 5% penicillin/streptomycin (Gibco) at 37°C in 5% CO2.
- Transfect cells with a plasmid encoding mVenus-tagged p27K- (kindly provided by Toshio Kitamura, University of Tokyo; Oki et al., 2014) using EndofectinMax (GeneCopoeia) following the manufacturer's instructions.
- Serum starve cells for 48h. Use untransfected NIH/3T3 cells starved for 48h as control.
- Fix cells in 3.7% paraformaldehyde (in PBS) for 15min at 4°C followed by incubation in 0.3% Triton X-100 for 10min.
- Block unspecific binding sites in PBT (phosphate buffered saline (PBS) containing 1% bovine serum albumin, 0.5% Tween-20) for 1h at RT.
- Incubate cells with primary mouse anti-P27 antibody (antibodies-online, ABIN3025539, lot V2438-171009) diluted 1:100 in PBS ON at 4°C.
- Wash cells with TBST (50mM Tris-HCl, pH7.4, 150mM NaCl, 0.1% Tween 20) for 15min.
- Incubate cells with secondary antibody goat anti-mouse IgG (H+L) Alexa Fluor 555 (Invitrogen, A21422, lot 948498) diluted 1:1000 in PBS and DAPI (4’,6-Diamidino-2-phenylindole; Sigma D-9542).
- Image acquisition on Zeiss LSM 510 confocal microscope and processing using Adobe Photoshop 5.0.
- Notes
Validation #102346 (Immunocytochemistry)![Testé avec succès 'Independent Validation' signe]()
![Testé avec succès 'Independent Validation' signe]() Validation Images
Validation Images![Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.]() Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.
Protocole
Different volumes of total cellular lysates of either mVenus-p27K- transfected NIH/3T3 cells (1, 2, 3) or untransfected NIH/3T3 cells (4, 5) were loaded and the blot membrane was incubated with ABIN3025539 followed by incubation with anti-mouse IgG-linked to HRP and chemiluminescence detection. Expected molecular weight of p27 fused to mVenus is approximately 55kDa.
Protocole -
- Concentration
- 0.2 mg/mL
- Buffer
- 0.2 mg/mL in 1X PBS with 0.1 mg/mL BSA (US sourced) and 0.05 % sodium azide
- Agent conservateur
- Sodium azide
- Précaution d'utilisation
- This product contains Sodium azide: a POISONOUS AND HAZARDOUS SUBSTANCE which should be handled by trained staff only.
- Stock
- 4 °C,-20 °C
- Stockage commentaire
- Store the p27 antibody cocktail at 2-8°C (with azide) or aliquot and store at -20°C or colder (without azide).
-
- Antigène
- P27
- Synonymes
- anticorps Kip1, anticorps p27, anticorps cyclin dependent kinase inhibitor 1B, anticorps CDKN1B
- Classe de substances
- Viral Protein
- Sujet
- Recognizes a 27 kDa protein, identified as the p27Kip1, a cell cycle regulatory mitotic inhibitor. Its epitope spans between aa 83-204 of p27. It is highly specific and shows no cross-reaction with other related mitotic inhibitors. p27Kip1 functions as a negative regulator of G1 progression and has been proposed to function as a possible mediator of TGF- induced G1 arrest. p27Kip1 is a candidate tumor suppressor gene. This mAb co-precipitates cdk4 in complex p27Kip1 and is excellent for staining of formalin-fixed tissues.
-

 (2 validations)
(2 validations)




