Lapin anti-Chévre IgG Anticorps (Colloidal Gold)
-
- Antigène Tous les produits IgG
- IgG
-
Reactivité
- Chévre
-
Hôte
- Lapin
-
Clonalité
- Polyclonal
-
Conjugué
- Colloidal Gold
-
Application
- Immunohistochemistry (Frozen Sections) (IHC (fro)), Immunohistochemistry (Paraffin-embedded Sections) (IHC (p)), Immunoelectron Microscopy (IEM)
-
-
- Indications d'application
- Optimal working dilutions should be determined experimentally by the investigator.
- Restrictions
- For Research Use only
-
- by
- Okeanos Research Laboratory, Department of Biological Sciences, Clemson University
- No.
- #103646
- Date
- 18.10.2016
- Antigène
- goat IgG
- Numéro du lot
- Application validée
- Electron Microscopy
- Contrôle positif
- goat anti-CBD-SNAP primary antibody
- Contrôle négative
- no primary control
- Conclusion
Passed. ABIN1720731 specifically binds to primary antibody raised in goat.
- Anticorps primaire
- goat anti-CBD-SNAP
- Anticorps secondaire
- ABIN1720731
- Full Protocol
- Oyster visceral mass tissue is dissected and fixed in 4% paraformaldehyde in seawater ON.
- Sections is rinsed with 70% ethanol and then transferred to 70% ethanol for about 2h at RT before automated dehydration process.
- Serial dehydration process using an automated ASP300S Enclosed Tissue Processor (Leica Biosystems) as follows:
- 70% ethanol for 45min
- 90% ethanol twice for 45min
- 100% ethanol twice for 45min
- xylene twice for 45min
- paraffin wax at 58°C 3 times for 30min
- Tissue is mounted in a paraffin block and hardened ON.
- 8µm tissue sections are retrieved from the block and collected on circular glass cover slips.
- Heat cover slips at 60°C for 1h.
- Deparaffination and rehydration:
- Xylene twice for 15min
- 100% ethanol twice for 10min
- 95% ethanol for 10min
- 85% ethanol for 10min
- 70% ethanol for 10min
- 50% ethanol for 10min
- 30% ethanol for 10min
- distilled water for 10min
- PBS for 10min
- Wash tissue sections with PBS with 0.05% triton X twice for 30min.
- Permeabilize in PBS with 0.05% triton X ON.
- Treatment of the tissue sections with 1mg/mL sodium borohydride in PBS three times for 5min to reduce autofluorescence.
- Wash sections in PBS 3 times for 15min for at RT.
- Block sections in PBST with 1% BSA for 2h at RT.
- Incubate sections with goat anti-CBD-SNAP antibody (lab stock) diluted 1:200 in PBST with 1% BSA ON at 4°C to detect the location of chitin.
- Wash sections in PBS 3 times for 15min with PBS at RT.
- Additionally, incubate the CBD-SNAP and SNAP-tag double-stained sections with rabbit anti-SNAP antibody (antibodies-online, ABIN1573927, lot 13D000621) diluted 1:200 in PBST with 1% BSA ON at 4°C.
- Wash sections in PBS 3 times for 15min at RT.
- Incubate sections with the secondary rabbit anti-goat IgG (whole molecule) antibody Colloidal Gold (5nm) conjugate (antibodies-online, ABIN1720731) diluted 1:50 in PBST with 1% BSA in the dark ON at 4°C.
- Wash sections in PBS three times for 15 min at RT.
- Post fix sections with 1% glutaraldehyde in PBS for 15min at RT.
- Wash sections in distilled water three times for 10min at RT.
- R-Gent SE-EM silver enhancement reagents (Aurion, 2551, lot 60323/2):
- Prepare a slower developer mix with 1:60 ratio of initiator: activator.
- Silver enhancement for 30 min with 1:20 developer: enhancer ratio.
- Stop the reaction by washing with distilled water for 5 min 3 times.
- Graded alcohol series:
- 50% alcohol for 15min
- 70% alcohol for 15min
- 85% alcohol for 15min
- 95% alcohol for 15min
- 100% alcohol for 15min
- Critical point drying of the sections with CO2 transitional fluid for 15min.
- Slowly degas sections at a degassing rate of 100Pa/min.
- Sections are mounted on the same aluminum stub, coated with carbon and visualized at 20kV with probe current at 40, with basic SEM-BSE mode on a Hitachi S-3400 Variable Pressure SEM, working distance kept at 10mm. Brightness and contrast settings are kept constant for the both samples.
- Notes
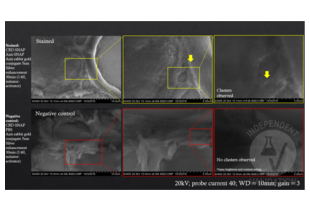
To validate the specificity of ABIN1720731, 8µm paraffin sections of oyster’s visceral mass were observed in this study. We compared the level of silver aggregating ability in the presence and absence of ABIN1720731, using commercially available silver enhancement method and SEM-BSE imaging. We found that the test samples treated with ABIN1720731 showed distinctive silver aggregates while the primary goat produced anti-CBD-SNAP antibody (ABIN1573927) was present. Such aggregates were not observable in the negative control sample without the primary antibody.
Validation #103646 (Electron Microscopy)![Testé avec succès 'Independent Validation' signe]()
![Testé avec succès 'Independent Validation' signe]() Validation Images
Validation Images![SEM-BSE images of oyster visceral mass tissue stained with CBD-SNAP and anti-SNAP primary antibodies and anti-Rabbit IgG (Whole Molecule) secondary antibody (Colloidal Gold (5nm)) ABIN3042038 and subjected to silver enhancement were imaged in ascending magnification (Top row). As a result clusters of silver aggregates were detected. In the absence of Anti-SNAP (Bottom row), the negative control did not show any signs of silver aggregation suggesting specificity of ABIN3042038 to the presence of rabbit IgG. Images in ascending magnification from left to right.]() SEM-BSE images of oyster visceral mass tissue stained with CBD-SNAP and anti-SNAP primary antibodies and anti-Rabbit IgG (Whole Molecule) secondary antibody (Colloidal Gold (5nm)) ABIN3042038 and subjected to silver enhancement were imaged in ascending magnification (Top row). As a result clusters of silver aggregates were detected. In the absence of Anti-SNAP (Bottom row), the negative control did not show any signs of silver aggregation suggesting specificity of ABIN3042038 to the presence of rabbit IgG. Images in ascending magnification from left to right.
Protocole
SEM-BSE images of oyster visceral mass tissue stained with CBD-SNAP and anti-SNAP primary antibodies and anti-Rabbit IgG (Whole Molecule) secondary antibody (Colloidal Gold (5nm)) ABIN3042038 and subjected to silver enhancement were imaged in ascending magnification (Top row). As a result clusters of silver aggregates were detected. In the absence of Anti-SNAP (Bottom row), the negative control did not show any signs of silver aggregation suggesting specificity of ABIN3042038 to the presence of rabbit IgG. Images in ascending magnification from left to right.
Protocole -
- Conseil sur la manipulation
- Do not freeze.
- Stock
- 4 °C
-
- Antigène
- IgG
- Abstract
- IgG Produits
- Classe de substances
- Antibody
-

 (1 validation)
(1 validation)



